
The final filling process is a critical process step involving finished product filling, room-to-room transfer, sterilizing filtration, and aseptic filling. In this complex landscape, a single-use aseptic filling system emerges as a pre-validated, pre-assembled, and pre-sterilized production unit. Comprising single use liquid storage systems, wall-through systems, sterilizing filtration and receiving bags, filling bags, filling tubing, and single-use filling needles, they all offer efficiency and sterility.
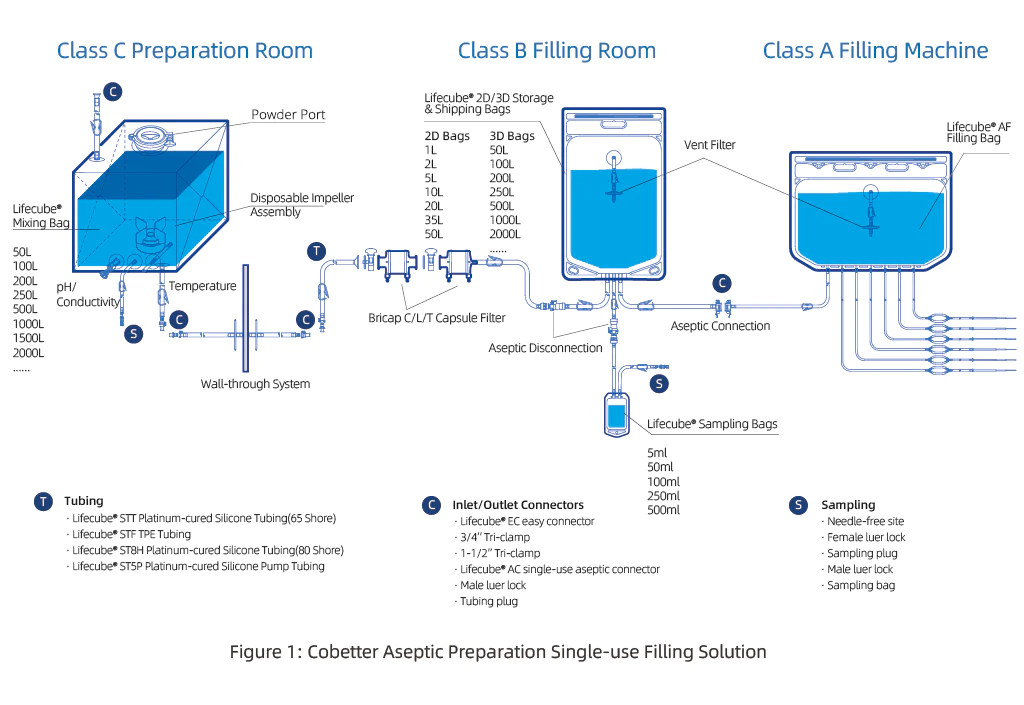
The success of a single-use aseptic filling system hinges on aligning the production environment with specific application requirements. For instance, the production of buffer bags in filling machines should adhere to a Class 100 (ISO 5, Class A) environment. Furthermore compliance with general regulatory requirements, including particulate matter control, extractables and leachables (E&L) evaluation, bacterial endotoxins, and sterility, is imperative.
Design considerations for single-use filling systems in diverse biopharmaceutical filling environments, such as laminar flow, O-RABS, C-RABS, and isolators, vary significantly. Key considerations include:
Cobetter’s single-use technology team plays a pivotal role in verifying and defining the filling accuracy and optimal recommended service life of filling tubes under worst-case conditions for various filling volume ranges and process times. This meticulous approach ensures the reasonable and stable use of single-use filling systems, promoting both efficiency and reliability.
Cobetter’s single-use aseptic filling system seamlessly integrates with common filling machine brands such as Bosch and Romaco. Tailored for vials, ampoules, pre-filled syringes, and other forms, it addresses the filling requirements of 1-12 needles. By negating the need for co-line risk assessments and cumbersome cleaning validations, Cobetter’s system enhances personnel protection levels, especially for highly toxic and active products.
Choosing a single-use aseptic filling system is not just a decision; it’s a strategic investment in the efficiency, sterility, and safety of biopharmaceutical production. Cobetter’s innovative approach, meticulous design considerations, and commitment to verification ensure that pharmaceutical companies can navigate the complexities of the formulation filling process with confidence and precision. The result is a streamlined and reliable production process that meets the highest industry standards.